by Arjun Walia, The Pulse:
 During a time of a huge loss of trust in institutions, you’d think companies would want to be more honest and forthcoming. Then again, does that even benefit them?
During a time of a huge loss of trust in institutions, you’d think companies would want to be more honest and forthcoming. Then again, does that even benefit them?
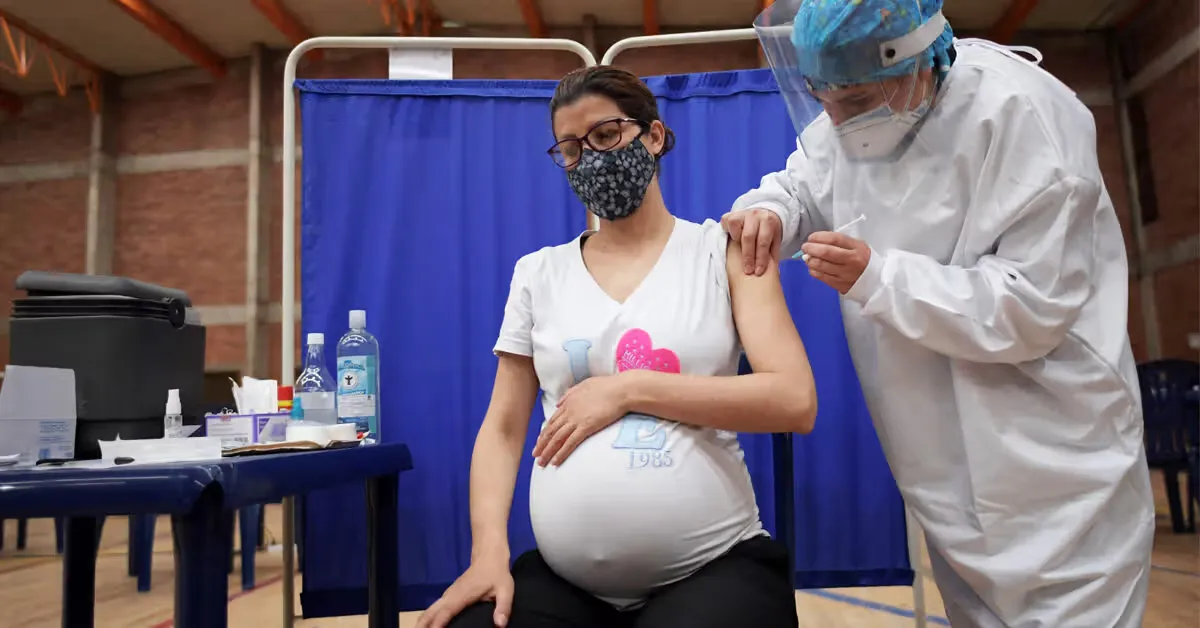
During the rollout of COVID vaccines, a lot of concerns were raised regarding the safety of the jab for pregnant women.
These concerns were drowned in a number of statements made by health authorities. In April 2021, four months after the COVID vaccine was granted emergency use authorization, CDC director Rachel Walensky recommended that all “pregnant people” get the vaccine. The American College of Obstetricians and Gynecologists (ACOG) “enthusiastically” recommended it three months later.
TRUTH LIVES on at https://sgtreport.tv/
These recommendations came despite the fact that there was zero evidence showing that the vaccine was safe for pregnant women.
In an attempt to settle the controversy, Pfizer launched a randomized controlled trial of 4,000 pregnant women. But just five months into the study, they stopped recruiting after 349 women had already been enrolled.
Pfizer never really explained why, and the most concerning fact is that the pregnancy outcomes of those who participated in the trial, and their babies, are still not public.
This isn’t a surprise, however. A 2021 study by Yale, Stanford, and the University of Pennsylvania published in The BMJ found that out of 58 new drugs that the FDA approved in a two-year period, 33% did not make their trial results public.
But given the extreme controversy around COVID vaccines, you’d think Pfizer would want to be transparent. Then again, they needed a court order to push them to be transparent with their trial results after wanting to wait 75 years before releasing them.
Pfizers Response
In 2022, Jelena Vojicic, vaccine medical lead at Pfizer Canada stated the following in an email that was released as a result of a court case,
“The study enrolment was stopped with incomplete numbers because recruitment was slow and it became unreasonable/inappropriate to randomize pregnant women to placebo given the amount of observational evidence that the vaccine is safe and effective, coupled with increasing number of technical committees supporting immunization of pregnant women”
Pfizer told investigative medical journalist Maryanne Demasi that,
“With the declining enrolment, the study had insufficient sample size to assess the primary immunogenicity objective and continuation of this placebo controlled study could no longer be justified due to global recommendations.”
So basically, women don’t need to know and there is no evidence according to Pfizer. Pfizer thought with slow enrolment it was unethical and dangerous to have a placebo group and not vaccinate pregnant women during a “deadly pandemic.”
The medical industrial complex had already declared it safe and effective regardless of any data. Why continue a trial if the vaccine is already being widely accepted as safe and effective for pregnant women? This is “groupthink science”, as Dr. Marty Makary, M.D., M.P.H refers to it.
This reminds of of an article I published in April regarding scientific consensus and what it actually means. You can read that here if interested.
Other Doctors Respond
Dr. Makary covered this story well in an article for the Tablet.
“They just recommended it for pregnant women with zero data. And maybe that’s why [Pfizer] stopped the study….running a study runs the risk of showing that there may not be a benefit in pregnant women.”
Tracy Beth Høeg MD, PhD, asked what many others did as well,
“Why? Do they not have enough money to fund the full trial? Lol. No, that’s not it. Could it be they found a concerning signal and did not want to chance letting it become significant? Hmmm. That sounds more likely.”
She then points out that 5.1% of infants of vaccinated moms had adverse events of special interest (AESI) between birth and 6 months while only 1.3% of infants in the control arm had AESIs.
“Because they limited the size of the trial, Pfizer can always say they didn’t find a significant signal. But if they don’t look, how can they find it?
Please note we don’t know what these adverse events were. They also didn’t report the birth outcomes (like stillbirths). Pfizer also didn’t report any efficacy data.
Why was the CDC “so sure” the mRNA covid vaccines were safe and effective in pregnancy? The answer is the same as it ever was: either they were basing their guidelines on feelings instead of data or, even worse, they were simply repeating what they were told to say.”
It would be nice to know exactly what these adverse reactions were. So far, COVID vaccines have made up a staggering majority of vaccine injury reports compared to all other vaccines combined over the past few decades.



